 The heaviest chalcogen, polonium, was isolated after an extraordinary effort by Marie Curie. To produce an image on paper, negatively charged toner particles are attracted to the positive image, transferred to a negatively charged sheet of blank paper, and fused with the paper at high temperature to give a permanent image. However, due to its extreme rarity and short half-life, the titel is often given to cesium. Dark areas in the original block the light and generate an invisible, positively charged image. Technically, the most reactive group 1 element is francium. Because amorphous selenium is a photosensitive semiconductor, exposing an electrostatically charged Se film to light causes the positive charge on the film to be discharged in all areas that are white in the original. Note that there is no clear division between metallic and non-metallic character.\): The Chemistry of Photocopying. Fluorine gas reacts explosively with many other elements and compounds, and is considered to be one of the most dangerous known substances. Atoms share electrons and form covalent bonds to satisfy the octet rule. 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. The atoms in group 6A make two covalent bonds. There are total 18 different groups in Periodic table. Non-metallic tendency increases going from left to right across the periodic table. Covalent bonds are formed by two atoms sharing electrons. Metallic tendency increases going down a group. Non-metallic character relates to the tendency to accept electrons during chemical reactions. 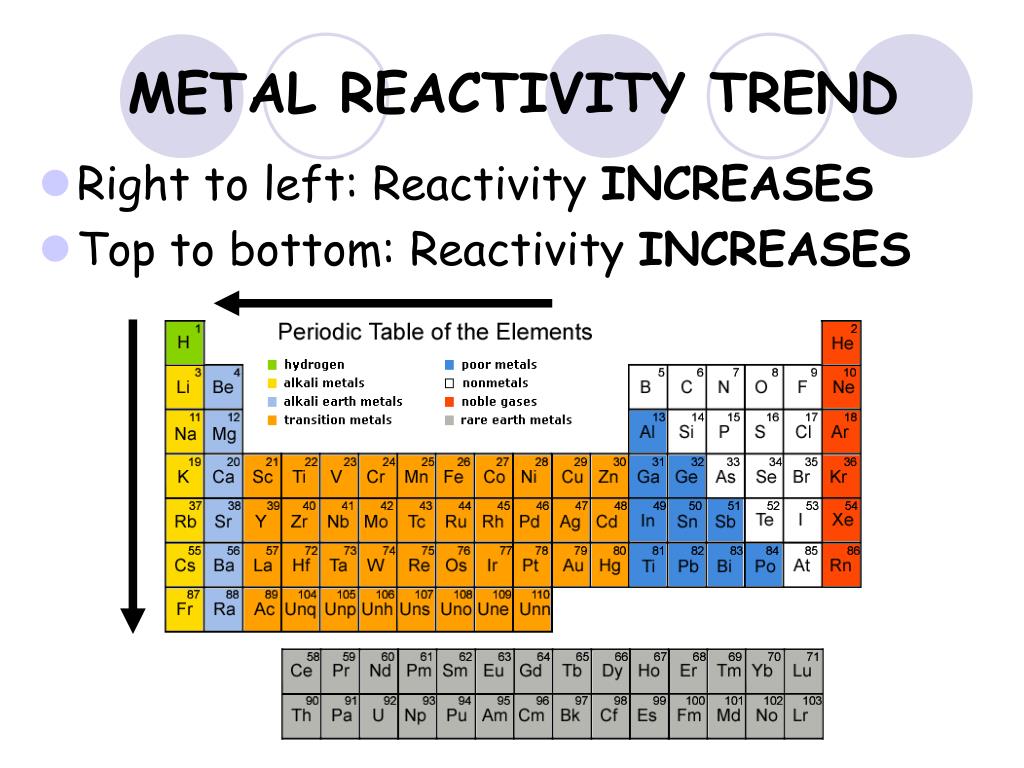
It is not found in nature as a free element. Metallic character refers to the level of reactivity of a metal. How many groups are in the periodic table There are 18 groups in the periodic table, one per each column of the periodic table. Elements in the same group have similar chemical properties. Part of Chemistry (Single Science) Atomic structure and the periodic table. 
Since the noble gases are a special group because of their lack of reactivity, the element fluorine is the most reactive nonmetal. The group with helium at the top of the periodic table is the least reactive.The periodic table is arranged in order of increasing atomic number. The Group 1 elements, also known as the alkali metals, all react vigorously with water to produce an alkaline solution. The most reactive nonmetals reside in the upper right portion of the periodic table. They are used in lamps, lasers, inert atmospheres, and balloons. They have a complete valence shell, high ionization energies, low boiling points, and are nearly inert. They are group 18, located in the right column of the table. 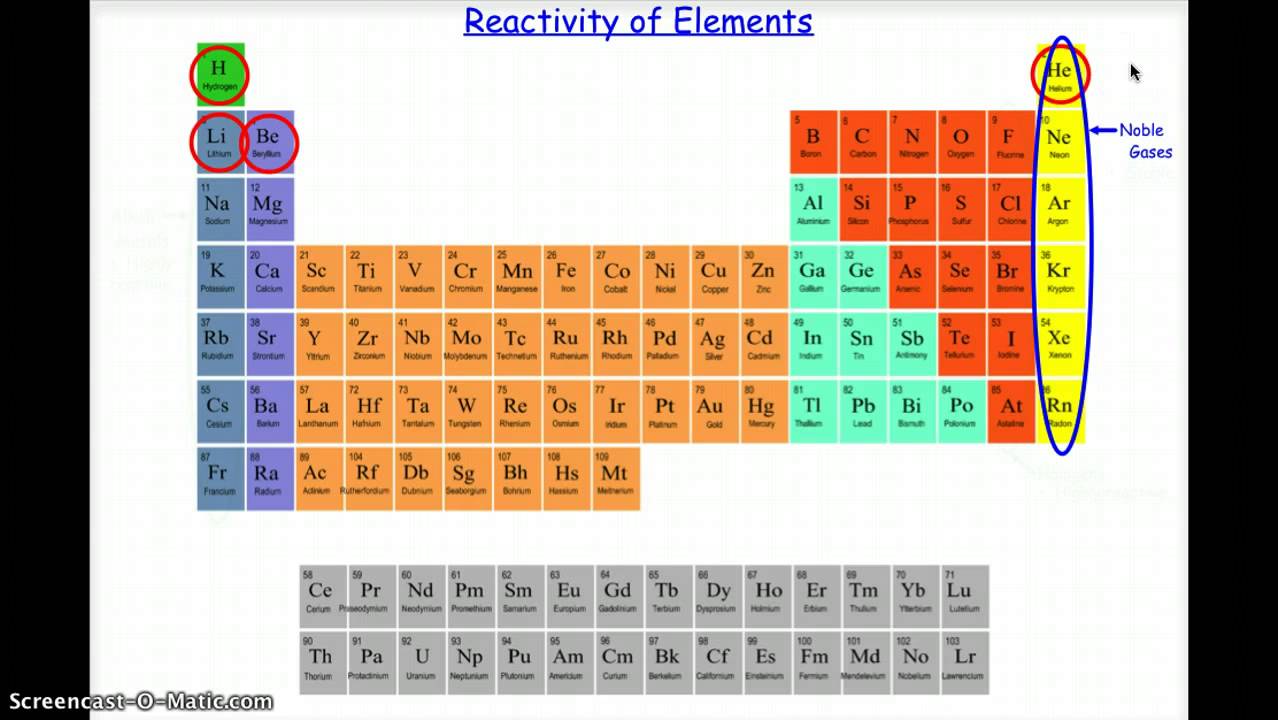
The least reactive elements, or most stable, on the Periodic Table are the Noble Gases (Group 8A) because they have octets (or 8 electrons) in their outermost electron shells. The noble gases are the least reactive chemical elements on the periodic table. As you can see, the pattern or trend moves more or less in an inside-out fashion. Nonmetals tend to gain electrons in chemical reactions, and have a high attraction for electrons within a compound. See below for a chart of reactivity in the Periodic Table. In addition, the atomic radius increases going down a group, placing the outer electrons further away from the nucleus and making that electron less attracted by the nucleus. Since the ionization energy decreases going down a group (or increases going up a group), the increased ability for metals lower in a group to lose electrons makes them more reactive. The metallic character increases as you go down a group. Reactivity of metals is based on processes such as the formation of halide compounds with halogens, and how easily the element displaces hydrogen from dilute acids. The least reactive elements, or most stable, on the Periodic Table are the Noble Gases (Group 8A) because they have octets (or 8 electrons) in their outermost electron shells. (Credit: User:Mirek2/Wikimedia Commons Source: Commons Wikimedia, Periodic Trends(opens in new window) License: Public Domain) As in group 15, the reactivity of elements in group 16 decreases from lightest to heaviest. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
